Key Takeaways
Kybella is FDA-approved deoxycholic acid used for reducing moderate to severe submental fat in adults.
Informed consent forms must disclose marginal mandibular nerve injury risk (4% in clinical trials) and all side effects.
A comprehensive template ensures compliance with HIPAA documentation requirements and legal protection for your clinic.
Pabau’s digital forms feature stores encrypted consent records and automates pre-treatment checklist workflows.
A kybella template is more than paperwork-it is your clinic’s primary defense against liability and regulatory findings. Without a comprehensive kybella consent form, you expose your practice to malpractice claims, fail to properly document patient understanding of material risks like marginal mandibular nerve injury, and leave your staff without guidance on what to discuss before injection. This guide provides a free kybella template ready to download and customize, covering everything from informed consent declarations to aftercare protocols-the same structure your clinic needs to document patient understanding and protect your business.
Download Your Free Kybella Consent Form Template
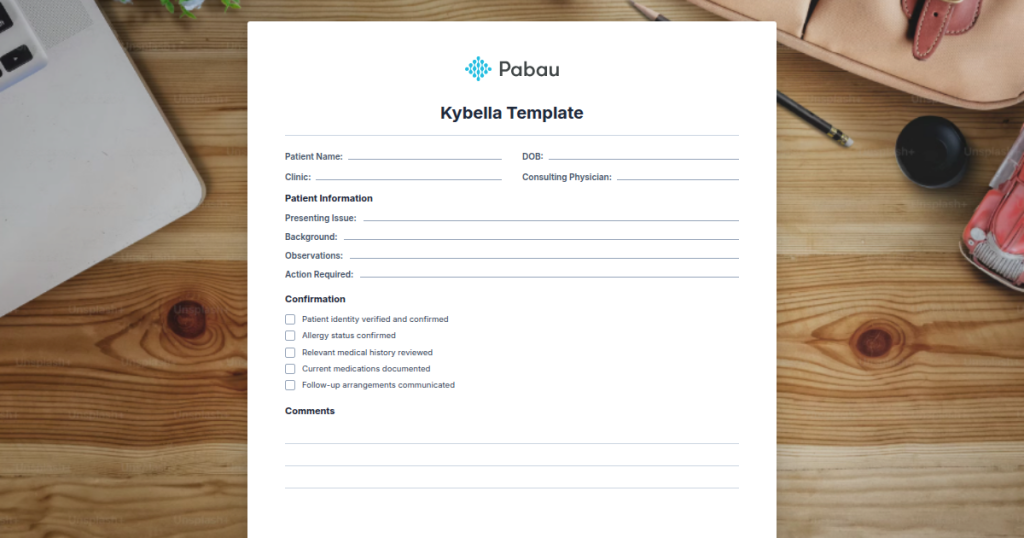
Kybella Consent Form
A ready-to-use informed consent form covering patient details, contraindication screening, risk disclosures for marginal mandibular nerve injury, deoxycholic acid mechanism of action, expected outcomes, side effects, pre-treatment assessment, and detailed aftercare instructions.
Download templateWhat is a Kybella Consent Form Template?
A Kybella consent form template is a clinical documentation tool that ensures patients understand the risks, benefits, and post-treatment expectations of deoxycholic acid injection therapy. Kybella is FDA-approved specifically for improvement in the appearance of moderate to severe convexity or fullness associated with submental fat (double chin) in adults. The consent document serves three critical functions: it demonstrates informed consent (legal requirement), discloses material risks including marginal mandibular nerve injury, and documents the patient’s acknowledgement before treatment begins.
Under HIPAA regulations, all aesthetic treatment consent forms must be retained as part of the patient’s medical record for a minimum of six years. A comprehensive facial consent form template structure protects both the patient and your clinic by creating a documented record of informed decision-making. Without it, a complication or patient complaint can escalate into a regulatory investigation or malpractice claim.
The template includes sections addressing deoxycholic acid’s mechanism (permanent fat cell destruction via cytolysis), treatment site anatomy (submental region and proximity to the marginal mandibular branch of the facial nerve), realistic outcome timelines, and contraindications for specific patient populations.
How to Use the Kybella Template in Your Clinic
The kybella template is structured around five operational steps that mirror your actual patient workflow:
- Patient Information Collection: Capture full name, date of birth, contact details, and emergency contact. Link this to your digital consent forms to populate fields automatically from your patient intake system.
- Contraindication Screening: Document absolute contraindications (pregnancy, allergy to deoxycholic acid, active infection at injection site, patient age under 18 as Kybella is FDA-approved for adults only) and relative contraindications (anticoagulant use, unrealistic expectations). This tiered approach allows clinical judgment while creating a defensible record.
- Risk Disclosure: Present the key adverse event from clinical trials: marginal mandibular nerve injury occurred in 4% of trial subjects, with all cases resolving spontaneously (range 1-298 days, median 44 days). Include numbness, bruising, and swelling timelines.
- Treatment Details and Consent Declarations: Include 5-7 numbered consent statements the patient must acknowledge (e.g., “I understand deoxycholic acid permanently destroys targeted fat cells,” “I acknowledge the risk of asymmetric smile from nerve injury,” “I agree to follow all pre and post-care instructions”). Use automated clinical documentation to populate treatment date, injector name, and procedure summary.
- Signature and Attestation: Obtain patient signature, injector signature, date, and time. Store digitally with encryption for HIPAA compliance.
Automate Your Consent Workflow
Store encrypted Kybella consent forms alongside patient records. Pabau's digital forms feature routes completed consents directly to client records and generates automated pre-care reminders.

Who is the Kybella Template Helpful For?
The kybella template applies to any healthcare setting offering deoxycholic acid injections for submental fat reduction:
- Medical Spas and Aesthetic Clinics: The primary user base. Clinics offering Kybella alongside other injectables (Botox, filler) benefit from a dedicated consent form that addresses the specific mechanism and risks of fat-dissolving agents.
- Dermatology Practices: Many dermatologists incorporate Kybella into cosmetic treatment menus. A specialized template ensures clear documentation of this aesthetic use.
- Plastic Surgery Offices: Surgeons offering non-surgical fat reduction options use this template as a standalone informed consent or as a supplement to pre-operative assessments.
- Nurse Practitioner and Physician Assistant-led Clinics: Non-physician injectors require explicit oversight and clear documentation of medical director review, making a robust consent form a compliance necessity.
Benefits of Using a Kybella Template
Legal Protection: A detailed consent form creates a documented record that the patient was informed of material risks before agreeing to treatment. This record is your primary defense in any malpractice claim or regulatory investigation.
Compliance with FDA Requirements: Kybella labeling requires disclosure of the 4% marginal mandibular nerve injury rate and the fact that all observed cases resolved spontaneously. A template ensures you meet these disclosure obligations.
Workflow Efficiency: A standardized kybella template removes the guesswork from consent conversations. Staff can guide patients through the form, answer predefined questions, and ensure nothing is missed, reducing time per patient while improving documentation quality.
HIPAA Documentation Audit Trail: Digital consent forms timestamped and linked to patient records create an auditable trail for compliance reviews and compliance checklist assessments.
Staff Training Standardization: When all staff use the same template, training is streamlined. New aesthetic nurses or injectors know exactly which risks to discuss and which documentation fields must be completed.
Pro Tip
Document the injection grid pattern used during treatment. Record the number of vials, injection depth, and exact anatomic landmarks to reduce future disputes about treatment scope. Many clinics photograph the marked submental region pre-treatment as visual proof of the treatment area.
Pre-Treatment and Aftercare Instructions in Your Template
The kybella template must include specific pre-treatment and post-treatment guidance to reduce complications and set accurate patient expectations:
Pre-Treatment Requirements
Instruct patients to avoid blood thinners (aspirin, NSAIDs, ibuprofen) for 48 hours before injection. Discontinue anticoagulant medications per physician guidance. Avoid alcohol and caffeine 24 hours prior, as these increase bruising risk.
Post-Treatment Aftercare Protocol
Most patients experience swelling, bruising, numbness, and induration (hardness) in the injection area for 5-14 days. Instruct them to ice the area for the first 24-48 hours, apply heat thereafter, wear compression garments if tolerated, and avoid heat, sun exposure, and strenuous exercise for one week. Numbness may persist for weeks; provide reassurance it is temporary.
Schedule follow-up assessment at 2-4 weeks post-treatment to evaluate fat reduction and plan additional treatment sessions if desired. Patients may receive up to 6 treatment sessions spaced at least 1 month apart, with the exact number tailored to the amount of submental fat and aesthetic goals (59% of clinical trial subjects received all 6 treatments).
Key Medical Considerations for Kybella Consent
Deoxycholic acid is a naturally occurring bile acid that breaks down fat cell membranes, causing permanent cell death (cytolysis). Once destroyed, fat cells do not regenerate. However, weight gain can still occur in other areas of the face or body if the patient gains weight post-treatment.
Marginal Mandibular Nerve Injury: The most significant risk disclosed in your template. This nerve controls the muscles that lower the lower lip. Injury results in asymmetric smile or facial weakness. In clinical trials, 4% of patients experienced this complication, but all cases resolved spontaneously. Resolution time ranged from 1-298 days, with a median of 44 days.
Absolute Contraindications: Pregnancy (no fetal safety data), allergy to deoxycholic acid or any template ingredient, active infection at injection site, patient age under 18 (Kybella is FDA-approved for adults only and safety and effectiveness in patients under 18 have not been established), and bleeding disorders on anticoagulation therapy without clearance.
Patient Selection: Best candidates are adults with moderate to severe submental fullness who have realistic expectations and understand that results appear gradually (peak effects at 8-12 weeks, with continued improvement up to 6 months). Avoid patients with unrealistic timelines or those expecting a facelift-like transformation.
Continue your research
Need guidance on informed consent best practices? Cosmetic Surgery Consent Form outlines the core elements every aesthetic procedure requires, from contraindication screening to risk disclosure to signature protocols.
Looking to streamline your consent workflow? Medical Spa Software integrates digital consent forms with patient records, automating pre-care reminders and ensuring every form is signed and time-stamped before injection.
Protect Your Clinic with a Comprehensive Kybella Consent Template
A detailed kybella template is not optional-it is the foundation of compliant, defensible aesthetic practice. Without it, you risk regulatory findings, malpractice claims, and patient disputes that could have been prevented with proper documentation. The template provided here covers informed consent, marginal mandibular nerve injury disclosure, realistic outcome timelines, contraindication screening, and detailed aftercare-everything your clinic needs to ensure patients understand risks and your team has a clear record of that understanding.
Download the template today, customize it to your clinic’s specific protocols, and integrate it into your digital workflow using Pabau’s digital forms system. Your patients will appreciate the professionalism, and your clinic will benefit from the documented compliance record.
Frequently Asked Questions
Kybella (deoxycholic acid) is FDA-approved for reducing moderate to severe submental fat (double chin) in adults. The active ingredient destroys fat cells permanently through a process called cytolysis.
Patients may receive up to 6 treatment sessions spaced at least 1 month apart, with the exact number tailored to the amount of submental fat and aesthetic goals (59% of clinical trial subjects received all 6 treatments). Sessions continue until the patient achieves their desired contour or fat reduction plateaus.
Marginal mandibular nerve injury is damage to the nerve controlling lower lip muscles, resulting in asymmetric smile or facial weakness. Your template must disclose that this occurred in 4% of clinical trial subjects, with all cases resolving spontaneously in 1-298 days (median 44 days).
Patients should avoid heat, sun exposure, and strenuous exercise for one week post-treatment. Swelling and bruising typically resolve within 5-14 days. They can return to normal daily activities immediately but should limit physical exertion.
Include patient demographics, informed consent declarations addressing key risks, contraindication screening, signatures from patient and injector with date and time stamps, and secure digital storage. This creates an auditable record of informed decision-making.